60 GWh of Sodium-Ion. One Big Question.
On 27 April 2026, CATL and HyperStrong signed a three-year cooperation agreement for 60 GWh of sodium-ion battery cells. That is the largest sodium-ion order ever placed, equivalent to half of all energy storage batteries CATL delivered in 2025.
The specs on paper are impressive (source: CATL-HyperStrong press release via PR Newswire, April 27, 2026):
• Cell capacity exceeding 300 Ah
• Energy density of around 160 Wh/kg
• System energy efficiency of 97%
• Cycle life of more than 15,000 cycles at 80% capacity retention
• Operating temperature range of -40°C to 70°C
At Sinovoltaics we have inspected both CATL’s and HyperStrong’s production lines. Good integrators, serious operations. And the deal makes strategic sense. Sodium is roughly 1,000 times more abundant than lithium, cheaper to source, and the cells can be fully discharged and transported at 0 V, which simplifies logistics compared to lithium-ion cells that must maintain a minimum voltage during shipping. A deal of this scale is paving the way for sodium-ion to deliver on its promise as the cost-efficient chemistry the grid storage market has been waiting for.
But one claim keeps appearing in every press release and investor deck: “operates at -40°C.” That claim deserves a closer look. Because operating and operating efficiently are not the same thing.
What “Operates at -40°C” Actually Means
CATL’s own data supports the claim. The Naxtra passenger EV battery retains over 90% of its discharge capacity at -40°C and still delivers power at -50°C (source: CATL Naxtra press release). For the energy storage variant, the operating range is listed as -40°C to 70°C (source: CATL-HyperStrong press release, April 27, 2026). The cells can discharge in extreme cold. That part is not in dispute.
This is a real advantage over LFP, which shows reduced capacity at low temperatures and faces lithium plating risk during cold charging.
But the marketing claim measures only one dimension: can the cell deliver power? For a grid-scale BESS cycling daily for revenue, the question is: what is the round-trip efficiency at low temperatures, and how much money am I losing per cycle?
What Independent Research Actually Shows
A peer-reviewed study in the Journal of Power Sources (Rehm et al., 2025, “Comparing the electrical performance of commercial sodium-ion and lithium-iron-phosphate batteries”) provides independent, side-by-side test data. Researchers from TU Munich and RWTH Aachen tested three commercial 18650 cells: one LFP and two sodium-ion cells with layered oxide cathodes and hard carbon anodes, the same chemistry CATL uses.
Impedance Increases Far More Than LFP
Pulse resistance is how much the cell's voltage drops when current is suddenly applied. It is a direct measure of the cell's internal losses. According to Rehm et al. (Section 3.3, Fig. 3), between 45°C and 10°C the pulse resistance of both sodium-ion cells increased by 360% to 450%. The LFP cell increased by approximately 100%. At low SOC (below 30%), sodium-ion pulse resistance spiked a further 50% to 100% compared to mid-range SOC. LFP showed only 3% to 6% variation across the same SOC range. In practical terms, a BESS with 4x more internal resistance at cold temperatures will tend to increase energy losses significantly, directly reducing revenue.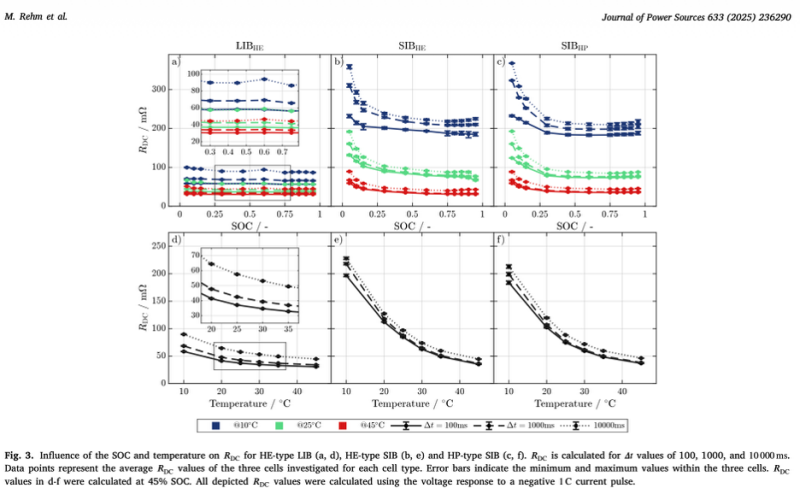
Efficiency Drops Hard Below 50% SOC
According to Rehm et al. (Section 3.5, Fig. 8), at 25°C and 0.67C, full-range round-trip efficiency (energy out divided by energy in) was 91.8% for the high-energy sodium-ion cell and 93.6% for the high-power variant. In the high SOC region (50-100%), efficiency rose to 94.6% and 95.6% respectively. In the low SOC region (0-50%), it dropped to 89.8% and 91.7%. Efficiency losses in the low SOC range were more than double those in the high SOC range.
LFP showed almost no SOC dependence: efficiency stayed between 92.6% and 94.4% regardless of SOC window (Rehm et al., Section 3.5).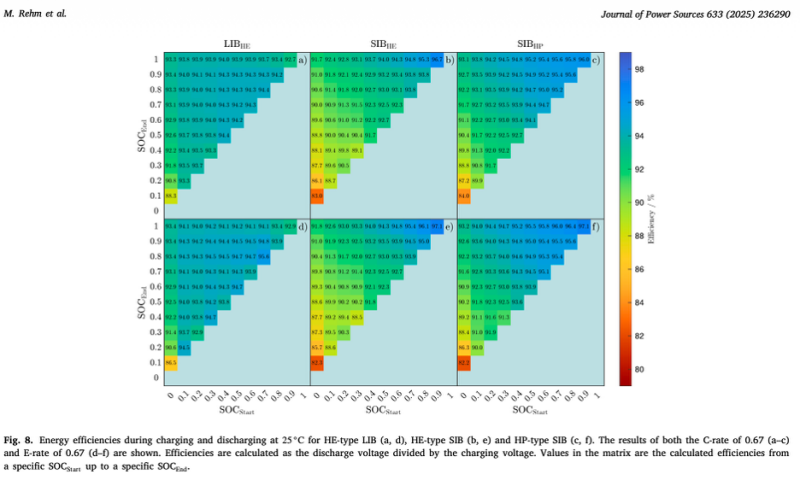
At 10°C, Efficiency Degrades Sharply Under High Load
Rehm et al. report that the high-energy sodium-ion cell’s energy efficiency was “drastically reduced at 10°C,” with coulombic efficiency dropping below 80% at 1C or higher (Section 3.4). Self-heating exceeded 20°C for the high-energy cell and 15°C for the high-power cell (Section 3.4, Fig. 6a-c). That is energy wasted as heat.
By comparison, the LFP cell retained significantly higher efficiency at 10°C across all load rates (Rehm et al., Fig. 6 d-f). The authors note that the SIBs’ “higher resistance and impedance values” and “stronger dependence on temperature” show up clearly in the efficiency data (Section 4).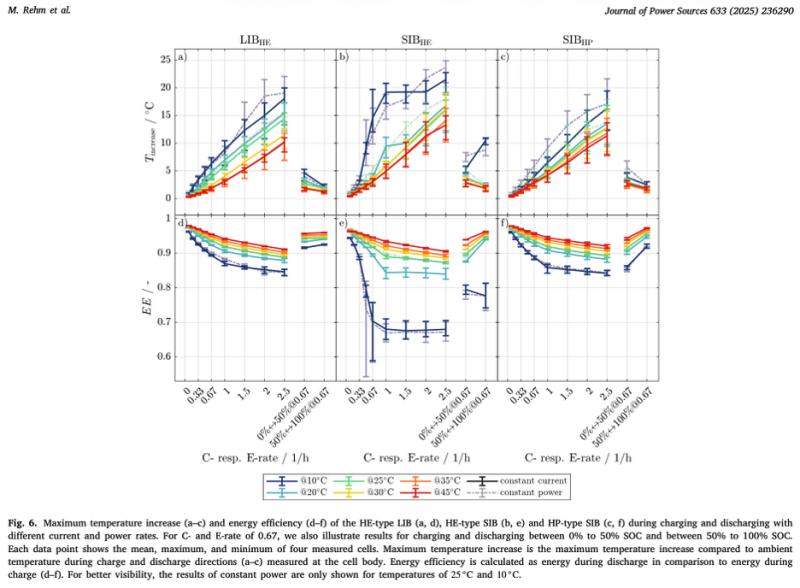
Sodium Plating: The Same Problem, Different Metal
By definition, sodium-ion avoids lithium plating. But Rehm et al. state that “the resistance measurements support the assumption that sodium plating occurs due to high overpotentials at low temperatures” on the hard carbon anode (Section 3.4). They reference Hijazi et al. (2024), who “have proven plating can occur for SIBs with layered oxides as cathode material and hard carbon as anode material.” Bai et al. (2024) confirm in Advanced Energy Materials that the hard carbon low-voltage plateau sits close to the sodium metal plating potential, making this worse at low temperatures and high charge rates.
Bai et al. (2024) go further in their review, stating that below -10°C, conventional SIBs experience “a substantial reduction in their satisfactory discharge capacity, rendering them virtually non-rechargeable.” Sodium-ion does not eliminate plating. It replaces lithium plating with sodium plating.
The Gap Between the Datasheet and the Site
CATL’s 97% system energy efficiency claim is plausible at 25°C, at moderate C-rates, in the upper SOC range. But the independent data from Rehm et al. paints a different picture at lower temperatures. I nonetheless expect CATL to have implemented design improvement not reflected in currently published independent data. At this scale, they almost certainly have. But until third-party data confirms it, the published research tells us:
• Coulombic efficiency drops below 80% at 10°C (Rehm et al., Section 3.4)
• Self-heating exceeds 20°C at 10°C ambient, loading the thermal management system (Section 3.4, Fig. 6)
A BESS in northern Europe, the US Midwest, or northern China will see temperatures below 10°C for months. None of the above appears on a product datasheet.
What This Means for Procurement
Sodium-ion has real advantages: lower material cost, no lithium or cobalt, safe 0 V transport, and good cycling at moderate temperatures. For warm-climate grid storage, it can be a strong choice.
But for cold-climate deployments, the risk profile changes.
Your FAT protocol needs updating. Testing at 25°C and 50% SOC will not catch the temperature-dependent and SOC-dependent behaviours that define sodium-ion. Test at 10°C across the full SOC range.
Your contract specs need realistic conditions. A 97% efficiency guarantee at 25°C is not the same as one at your site’s winter temperature.
Your thermal management needs rethinking. Rehm et al. measured over 20°C of self-heating at 10°C ambient (Section 3.4, Fig. 6). A system sized for LFP will be undersized for sodium-ion. This self-heating also increases the system's own energy consumption, which must be accounted for in self-consumption calculations and thoroughly tested during FAT.
The Bottom Line
“Operates at -40°C” is a discharge capability claim. It is not an efficiency claim. For a BESS cycling thousands of times per year, the difference between those two statements is where the margin goes.
Before you sign a sodium-ion BESS contract, make sure the performance guarantees match your site conditions, not the test lab conditions.
Planning a sodium-ion BESS project? Talk to our team about adapting your FAT protocol and acceptance criteria for sodium-ion chemistry. We have inspected HyperStrong’s production lines and can help you verify that what ships matches what was promised. Visit sinovoltaics.com/bess-inspection for details.
Sources
1. Rehm, M., Fischer, M., Rubio Gomez, M., Schuette, M., Sauer, D.U., Jossen, A. (2025). "Comparing the electrical performance of commercial sodium-ion and lithium-iron-phosphate batteries." Journal of Power Sources, 633, 236290. https://doi.org/10.1016/j.jpowsour.2025.236290
2. Hijazi, H. et al. (2024). "Overview of electrochemical competing process of sodium storage and metal plating in hard carbon anode of sodium ion battery." Energy Storage Materials. https://www.sciencedirect.com/science/article/abs/pii/S2405829724004719
3. Bai, Z. et al. (2024). "Low-Temperature Sodium-Ion Batteries: Challenges and Progress." Advanced Energy Materials. https://doi.org/10.1002/aenm.202303788
4. CATL press release: "CATL and HyperStrong Sign the World's Largest Sodium-Ion Energy Storage Cooperation Agreement." April 27, 2026. https://www.prnewswire.com/news-releases/catl-and-hyperstrong-sign-the-worlds-largest-sodium-ion-energy-storage-cooperation-agreement-302755769.html
5. CATL press release: "Naxtra Battery Breakthrough & Dual-Power Architecture." https://www.catl.com/en/news/6401.html