Nickel Cadmium Batteries
HVAC System
Stationary Energy Storage
Thermal Run Away
Standards for Lead-Acid Batteries
Standards for Lithium-ion Battery
LTO batteries
Solid-state Li-ion batteries
[...]
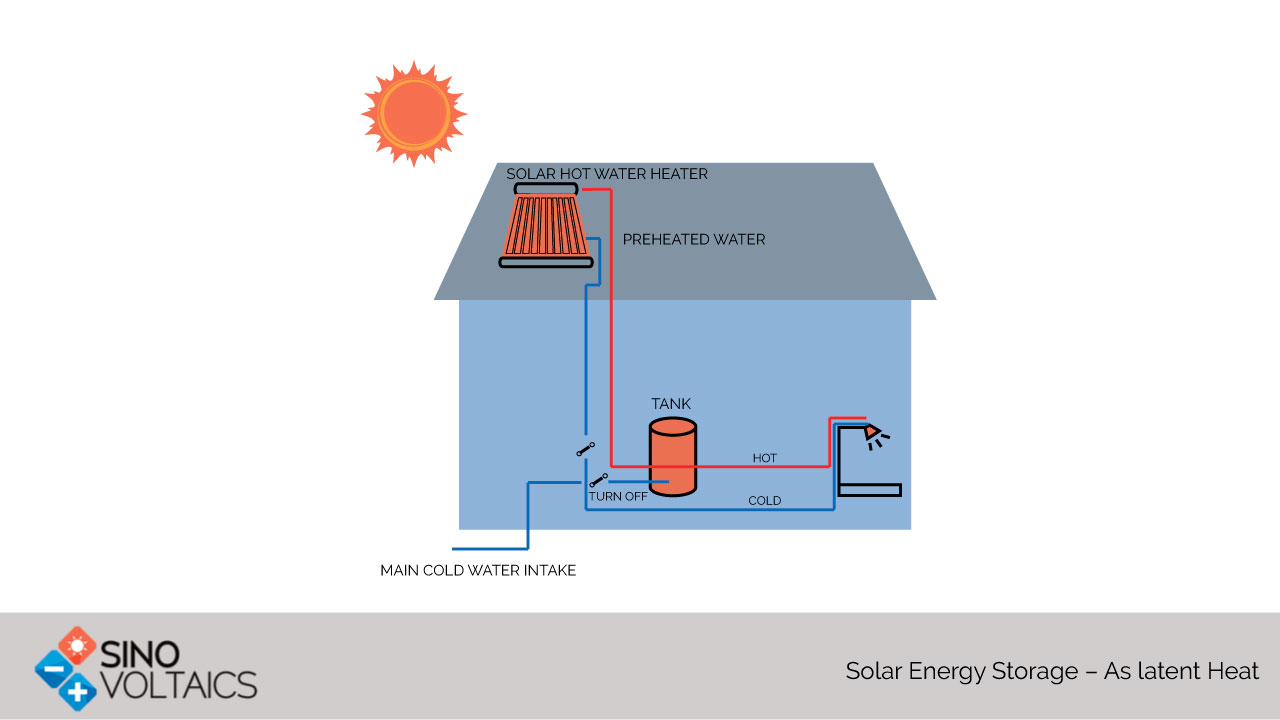 Phase Change Materials(Source: IAE 2005)Phase change heat storage works at relatively lower temperatures than sensible heat storage and involves a constant temperature or very little change. They are quite suited for temperature stabilization. A phase change above the ambient will store heat and releasable later. A phase change below the ambient temperature will store cold. Thus, they are relevant to HVAC operations. Heat storage can be in independent insulated heat and cold stores. Alternatively, and more popularly, heat storage can be integrated into the building structure by making PCMs part of cement, concrete and other construction material. Use in buildings contributes to a cleaner environment. Classification of Phase Change MaterialsThere are two broad classes of phase change materials - organic and inorganic.· Inorganic Phase Change Materials- Inorganic phase change materials were the earliest favorites in the search for phase change energy storage. Other than water and ice, the common favorite inorganic PCMs are salt hydrates. These materials are commonly available, low-cost, and non-inflammable. They have high latent heat which means higher energy density storage. The disadvantages include corrosiveness, tendency to supercool (reducing storage), improper solidification, and long-term instability. The disadvantages may be reduced by addition of small percentages of suitable gelling or thickening materials.· Organic Phase Change Materials- Organic phase change materials include paraffin and fatty acids. They have better chemical stability and do not suffer from supercooling effect. They are better integrated with building materials. The initial cost is higher than inorganic materials, but the installed costs are comparable. However, organic PCMs are inflammable, and can generate harmful fumes in case of fire. Some of them suffer from volume change over time, odor, thermochemical oxidative aging, and reaction with hydration products of concrete. Steam AccumulatorsBoth latent and sensible heat are stored in in “steam accumulators” which are high pressure tanks containing superheated steam and water. Feeding energy to the reservoir increases the percentage of steam and increases the pressure. Withdrawal of energy will cool the steam into water and lower the pressure. They work in conjunction with the boilers in a steam generation plant and are very helpful in peak shaving because of instant reaction.Ice StorageConverting water to ice will also store energy at the freezing point (0˚C). Ice will melt to provide cooling. This can be useful in air-conditioning of buildings, and cold storage of fruits and vegetables. An interesting fact is that in the face of a famine, Brigham Young of now Utah State, had used a freezing lake to store meet for his tribe by driving their cattle into the lake till they froze. Latent Heat Storage at High Temperatures-Metallic AlloysLatent heat storage at low temperatures is not favorable to power generation. For this, phase change materials which melt at high temperatures are required. Instead of using a fusible material inside a tank of another material an alloy of two materials with a reasonable miscibility gap i.e., difference in melting points is used. Heat is stored in the latent heat of fusion of the lower melting metal while the other helps retain shape. This technology has the advantage of higher energy density, stability over time, and high thermal conductivity to permit absorption and release of heat at higher rates.
Phase Change Materials(Source: IAE 2005)Phase change heat storage works at relatively lower temperatures than sensible heat storage and involves a constant temperature or very little change. They are quite suited for temperature stabilization. A phase change above the ambient will store heat and releasable later. A phase change below the ambient temperature will store cold. Thus, they are relevant to HVAC operations. Heat storage can be in independent insulated heat and cold stores. Alternatively, and more popularly, heat storage can be integrated into the building structure by making PCMs part of cement, concrete and other construction material. Use in buildings contributes to a cleaner environment. Classification of Phase Change MaterialsThere are two broad classes of phase change materials - organic and inorganic.· Inorganic Phase Change Materials- Inorganic phase change materials were the earliest favorites in the search for phase change energy storage. Other than water and ice, the common favorite inorganic PCMs are salt hydrates. These materials are commonly available, low-cost, and non-inflammable. They have high latent heat which means higher energy density storage. The disadvantages include corrosiveness, tendency to supercool (reducing storage), improper solidification, and long-term instability. The disadvantages may be reduced by addition of small percentages of suitable gelling or thickening materials.· Organic Phase Change Materials- Organic phase change materials include paraffin and fatty acids. They have better chemical stability and do not suffer from supercooling effect. They are better integrated with building materials. The initial cost is higher than inorganic materials, but the installed costs are comparable. However, organic PCMs are inflammable, and can generate harmful fumes in case of fire. Some of them suffer from volume change over time, odor, thermochemical oxidative aging, and reaction with hydration products of concrete. Steam AccumulatorsBoth latent and sensible heat are stored in in “steam accumulators” which are high pressure tanks containing superheated steam and water. Feeding energy to the reservoir increases the percentage of steam and increases the pressure. Withdrawal of energy will cool the steam into water and lower the pressure. They work in conjunction with the boilers in a steam generation plant and are very helpful in peak shaving because of instant reaction.Ice StorageConverting water to ice will also store energy at the freezing point (0˚C). Ice will melt to provide cooling. This can be useful in air-conditioning of buildings, and cold storage of fruits and vegetables. An interesting fact is that in the face of a famine, Brigham Young of now Utah State, had used a freezing lake to store meet for his tribe by driving their cattle into the lake till they froze. Latent Heat Storage at High Temperatures-Metallic AlloysLatent heat storage at low temperatures is not favorable to power generation. For this, phase change materials which melt at high temperatures are required. Instead of using a fusible material inside a tank of another material an alloy of two materials with a reasonable miscibility gap i.e., difference in melting points is used. Heat is stored in the latent heat of fusion of the lower melting metal while the other helps retain shape. This technology has the advantage of higher energy density, stability over time, and high thermal conductivity to permit absorption and release of heat at higher rates. 